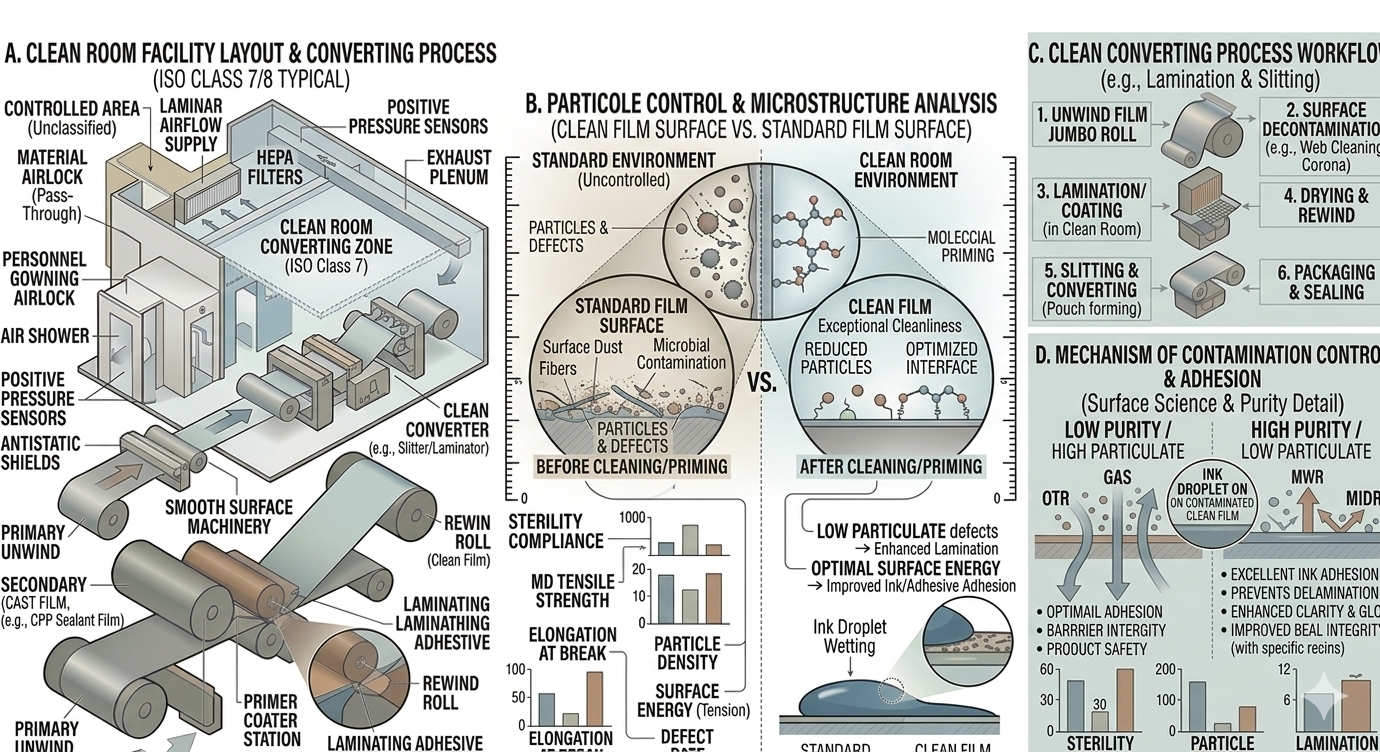
Definition
Clean room manufacturing refers to converting processes performed in controlled environments where airborne particles, temperature, and humidity are regulated to prevent contamination.
Applications
- Medical device component production
- Sterile barrier packaging
- Electronics and semiconductor materials
- Pharmaceutical packaging
Benefits
- Minimizes contamination risks
- Ensures product consistency
- Supports regulatory compliance
- Protects sensitive materials during manufacturing
Industry-specific examples
- Medical: cleanroom converting used to manufacture adhesive components for wearable medical devices.
- Industrial: controlled manufacturing environments used for contamination-sensitive sealing materials.
- Electronics: cleanroom processing used to convert films and laminates for semiconductor packaging.
Click to enlarge
FAQ
Q: Why are cleanrooms required in medical manufacturing?
A: They reduce airborne contamination that could compromise sterile or sensitive medical products.
Q: What standards define cleanroom classifications?
A: ISO 14644 standards classify cleanrooms based on allowable airborne particle levels.